A Polypoid Mass was Observed Crowing through the Left External Auditory Canal.
Luis Morejon-Troya12, Joyce Jimenez-Zambrano13
doi: http://dx.doi.org/10.5195/ijms.2019.341
Volume 7, Number 1: 11-14
Received 16 12 2018: Accepted 13 04 2019
ABSTRACT
IntroductionGlomus tumors are rare benign lesions of the middle ear that are often associated with symptoms of progressive hearing loss and pulsatile tinnitus. Diagnosis is often delayed due to the slow growth of the tumor, and surgical resections of the mass are the mainstay treatment.
Case PresentationA 56-year-old Latin American female was admitted to the hospital with a year-long history of intermittent pulsatile buzzing noise, progressive hearing loss, pain and discharge of the left ear. High-resolution Computerized Tomography and Magnetic Resonance Imaging revealed evidence of an enhancing mass lesion, which extended into the external auditory canal. A tympanomastoidectomy was performed. Most of the mass was removed, but some remained because of massive bleeding. The patient received 3 months of radiation therapy after the tympanic paraganglioma was surgically removed.
ConclusionThis case exposed the audiological findings, the location, and the extent of the tumor. Management options consist of radiation therapy and surgical management, depending on the size of the tumor. Early detection of glomus tumors is essential to prevent non-reversible complications.
Keywords: Glomus Tympanicum; Paraganglioma; Neuroendocrine Tumors; Paraganglioma; Extra-Adrenal; Neural crest-derived tumors (MeSH)..
Glomus tympanicum paragangliomas (GTPs), also called chemodectomas, are very uncommon neoplasms accounting for o.67o of all neoplasms in the head and neck and about o.03% of all tumors.1-3 They are neuroendocrine tumors that originate along the tympanic plexus associated with the tympanic branch of the glossopharyngeal nerve (Jacobson’s nerve) and the auricular branch of the vagus nerve (Arnold’s nerve) and are comprised of paraganglion cells derived from the neural crest.1-3
GTPs are soft and encapsulated tumors that are generally pulsatile when seen in vivo. The vascular nature of these tumors is evident histologically though the appearance of an extensive capillary network, leading to a reticular pattern under magnification. Though paragangliomas throughout the body appear to have the capacity for neurosecretory function, actively secreting TP are exceptionally infrequent, and the presence of elevated catecholamine markers should raise suspicion for a coexistent, separate abdominal or thoracic pheochromocytoma.4-5
GTPs are known to be locally aggressive but are rarely considered to be malignant. Destruction of the temporal bone and surrounding tympanic membrane is not unusually seen with paraganglioma growth. Once the middle ear space is saturated, GTPs may follow paths of least resistance and extend to the mastoid, Eustachian tube, or through the tympanic membrane and into the external auditory canal.6
Patients are most frequently female, and the age distribution was seen with peaks of around 20 and 60 years. Clinical findings are pulsatile tinnitus (81.4%), conductive hearing loss (77.1%) and aural fullness (70.2%). It is also common to observe in its early stages a blanching of the mass with application of positive pressure with pneumatic otoscopy. This finding is known as the Brown’s sign.7-8
High-resolution Computerized Tomography (CT) of the temporal bone is often initially used for lesions that appear to be limited to the tympanic cavity, with magnetic resonance imaging often complementary in more extensive lesions.9-10
We present a 56-year-old female patient with a tympanic paraganglioma that erodes through the tympanic membrane and extends through the auditory canal.
A 56-year-old Ecuadorian female, residing since her birth in a countryside area, was admitted to the hospital due to a year history of intermittent pulsatile buzzing noise, hearing loss, pain and discharge of the left ear.
Her past medical history included obesity, hypertriglyceridemia, hypercholesterolemia (2012), uterine myomatosis (2013) and fibromyalgia (2013). She had a surgical history of a cholesteatoma resection in the middle ear on the left side (2011) and a cesarean section 20 years ago. Her medications included amitriptyline 25 mg daily, buprenorphine 20 ug/h transdermal patch, acetaminophen 500 mg three times a day and vitamin B complex. She did not report any allergies. Her social history revealed occasional alcohol use in addition to being a current smoker of four cigarettes per day. She lived with her son. She had a family medical history of a mother with type 2 diabetes mellitus.
The first symptoms the patient referred started one year before admission. These symptoms were pulsatile buzzing noise and a feeling of left ear fullness. Six months after, she began to develop progressive left-sided facial droop and vertigo. She described all symptoms kept worsening until the day of admission to the hospital.
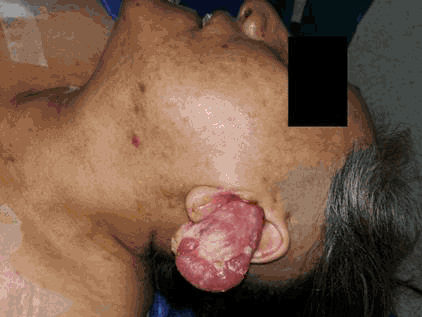
On physical examination, a pulsatile polypoid mass was observed growing through the left external auditory canal (Figure 1). Neurological examination revealed left hypoacusia and left-sided peripheral paresis of the facial nerve.
Figure 1.A Polypoid Mass was Observed Crowing through the Left External Auditory Canal.

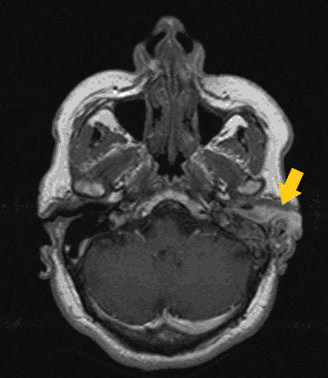
Pure tone audiometry revealed a severe conductive hearing loss with more than 60 dB air-bone gap (Figure 2) A magnetic resonance imaging (MRI) scan of the temporal bones demonstrated a mass occupying the cavum tympani, the mastoid air cells with extension to the left external auditory canal (Figure 3) The ossicles were missing, and the tumor mass engulfed the tympanic membrane.
Figure 2.Pure Tone Audiometry shows a Severe Conductive Hearing Loss on the Left Ear.

MRI shows Evidence of an Enhancing Mass Lesion which was Extending into the Left External Auditory Canal.
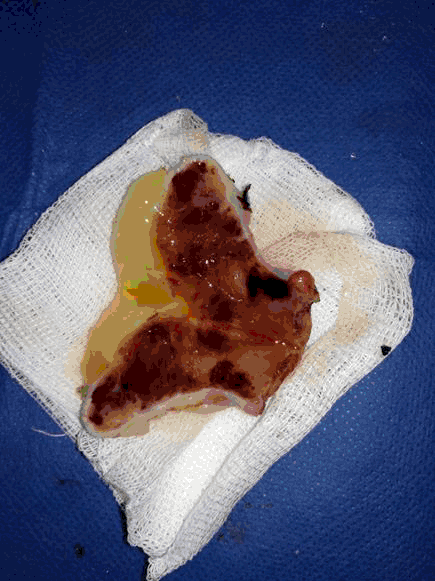
Finally, mastoidectomy was performed. After a retroauricular incision and open cavity tympanomastoidectomy, an irregular polypoid mass with a bleeding tendency was found, that filled the external auditory canal, middle ear cavity, and mastoid process. While the tympanic membrane was not localized, and auditory ossicles were disrupted, the sigmoid sinus, dura matter and facial canal were intact. Most of the mass was removed, but some remained because of massive bleeding (Figure 4)
Figure 4.A Gross Pathologic Sample of the Tympanic Paraganglioma. Notice the Friable Consistency of the Tumor.
On histologic examination, highly vascular tissue looking like granulation tissue was seen in a low power microscope field. In high power field, it revealed that the tumor was composed of nests of round to oval cells that were surrounded by delicate vascular septae resembling zellballen appearance. Findings on postoperative audiometry were unchanged from preoperative levels.
Our patient received a 3-month radiation therapy after the surgical removal of the tympanic paraganglioma. The patient was required for a follow-up after each radiation therapy. Despite all the efforts, unilateral hearing loss and peripheral facial nerve palsy persisted. Further, follow-up on the patient for a year has revealed no recurrence to date.
Most glomus tumors usually occur in adulthood with a female-to-male preponderance of 9:1. It frequently manifests as pulsatile tinnitus. Other complaints may include aural fullness, conductive hearing loss, and facial drooping on the affected side.3
GTPs occur along Jacobson nerve (a branch of the glossopharyngeal nerve) in the middle ear adjacent to the cochlear promontory.9 Grossly, tumors are solid and encapsulated, although they can have an invasive appearance in the temporal bone because of growth within the Haversian canal and air cells.5
Microscopically, tumors are conglomerations of chief cells surrounded by sustentacular cells (“zellballen”) and an extensive capillary network that creates a reticular appearance. Chief cells are characterized by a polyhedral shape, round nuclei and eosinophilic cytoplasm that can contain granular structures.11 Immunohistochemical staining generally reveals chromogranin, serotonin, neuron-specific enolase and somatostatin expression in chief cells and S-100 expression in sustentacular cells. Neurosecretory function in GTP tumor is rare, but screening for functional tumors remains an important part of paraganglioma management, if a patient is found to have elevated catecholamine level, they should be screened for other extra-adrenal paragangliomas and pheochromocytomas.12-13
All patients suspected of having GTP should undergo high-resolution temporal bone CT and potentially neck CT with contrast. The primary purpose of obtaining a temporal bone CT scan is to rule out a jugular paraganglioma with an erosion of the bone plate over the jugular bulb or other less common conditions such as an aberrant internal carotid artery, high riding jugular bulb, invasive intracranial tumors, encephalocele or facial nerve schwannomas with a dilated fallopian canal. A “salt and pepper” appearance may be visualized on T1- and T2-weighted MRIs, although this is an unusual finding. These hyper-and hypointense areas are related to slow- and fast-flowing intravascular blood and to intratumoral hemorrhage.10
In fact, the location of middle ear tumors may affect the clinical presentations of the patient, such as our case. The most common finding of a tympanic paraganglioma is a retrotympanic vascular mass (44.3%). It is rare for these tumors to grow through the tympanic membrane and into the external acoustic canal (13.9%).1 Hsu et al. showed a possible association between the presence of cholesteatoma and paraganglioma in the middle ear.3 To our knowledge, this would be the second reported case about the coexistence of glomus tympanicum and a previously diagnosed cholesteatoma.
Tumor stage is usually reported using the Glasscock-Jackson Staging System (Table 1). This system helps us to decide the general approach to the management of glomus tympanicum tumors. Some relative contraindications for surgical approach are advanced age (>65 years old), extensive skull-base or intracranial involvement, medical comorbidities (i.e. coagulopathies), and bilateral or multiple paragangliomas.14 While observation can be considered in patients with strong contraindications to surgery, gross total resection should remain the primary treatment modality for symptomatic patients who tolerate general anesthesia. A transcanal approach with a speculum is most commonly used for stage 1 disease. For stage 2, postauricular transcanal hypotympanotomy is utilized with greater frequency. Variations of intact canal wall tympanomastoidectomy are performed for stage 3 disease, and canal wall down with meatoplasty or canal closure is often performed in patients with stage 4 disease having an extensive external auditory canal tumor burden.11,13
Table 1.Glasscock-Jackson Classification14
| Grade | Definition |
|---|---|
| I | Small mass limited to the promontory |
| II | Tumor completely filling middle ear space |
| III | Tumor filling middle ear and extending into the mastoid |
| IV | Tumor filing middle ear, extending into the mastoid or through tympanic membrane fill the external auditory canal; may extend anteriorly to carotid |
The present case had an extensive tumor (stage 4) where surgery may increase the risk of neurological deficit; therefore, a subtotal resection with postoperative radiotherapy was indicated. The literature is limited for stage 4 glomus tympanicum. This is a rare case that shows us the natural course of the tumor and how aggressive it can be if it is not diagnosed in its course.
The differential diagnosis of tympanic paragangliomas depends significantly on the actual location of the tumor, such as the jugular and vagal paragangliomas. The differential diagnoses in tumors arising in the jugulotympanic area include middle ear adenoma, meningioma, schwannoma, and hemangiopericytoma, among others. Meningioma involving the jugular foramen can mimic the more common jugulotympanic paragangliomas.15-16
GTP is an important differential diagnosis when dealing with a patient that presents progressive hearing loss, pulsatile tinnitus and recurrent episodes of otitis media. This is because despite being a benign tumor, GTP has an aggressive growth, which in its natural course, can produce very severe destructions, as the present case. The imaging techniques are necessary to establish the extension, characteristics, destruction of adjacent structures. The CT is essential for establishing treatment and the surgical approach in these patients.
We would like to thank Dr. Zambrano and Dr. Cevallos for their guidance and support.
The Authors have no funding, financial relationships or conflicts of interest to disclose.
Conceptualization: LMT. Methodology: LMT. Resources: LMT, and JJZ. Writing – Original Draft: LMT, and JJZ. Writing – Review & Editing: JJZ. Supervision: LMT. Project Administration: LMT.
1.Portero de la Torre M, Sanchez Gutierrez R, Gallardo Muñoz I, Bravo Rodriguez FA. Paraganglioma timpánico con extension a trompa de Eustaquio y nasofaringe: A propósito de un caso. Acta Otorrinolaringol Esp. 2012;63(4):314–317.
2.Boedeker CC, Ridder CJ, Schipper J. Paragangliomas of the head and neck: Diagnosis and treatment. Fam Cancer. 2005;4(1):55–9.
3.Hsu JL, Hwang CF, Kao YF, Lui CC, Lin JW, Peng JP. Paraganglioma presenting as chronic otitis media with cholesteatoma: pitfalls and strategies. Am J Otolaryngol. 2004 May-Jun;25(3):190–194.
4.Bjõrklund P, Pacak K, Crona J. Precision medicine in pheochtomocytoma and pataganglioma: current and future concepts. J Intern Med. 2016 Dec;280(6):559–73.
5.Kumar V, Abbas A, Aster J. Robbins and Cotran Pathologic Basis of Disease. 9th ed. Elsevier; 2014.
6.Jethanamest D. Tympanic paraganglioma—Resection techniques. Oper Tech Otolayngol Head Neck Surg. 2016;27(1):15–19.
7.Hirunpat S, Riabtol K, Dechsukhum C, Atchariyasathian V, Tanomkiat W. Nasopharyngeal extension of glomus tympanicum: an unusual clinical and imaging manifestation. AJNR Am J Neuroradiol. 2006 Oct:27(9):1820–2.
8.Maurer CJ, Aschendorff A, Urbach H. Diagnosis of a tympanic paraganglioma with CT perfusion imaging: a technical note and case description. Neuroradiol J. 2017 Nov 24:197140091774457.
9.Lee JC, Malhotra A, Wang H, Westesson P-L. Paragangliomas of the Head and Neck: A Pictorial Essay. Univ Rochester Med Cent. 2007:1–25.
10.Kaylie DM, O'Malley M, Aulino JM, Jackson CG. Neurotologic surgery for glomus tumors. Otolaryngol Clin North Am. 2007 Jun;40(3):625–49.
11.Sennatoglu L, Sungut A. Histopathology of Paragangliomas. Otol Neurotol. 2002;23(1):104–5.
12.Carlson ML, Sweeney AD, Pelosi S, Wanna GB, Glasscock ME, Haynes DS. Glomus tympanicum: A review of 115 cases over 4 decades. Otolaryngol – Head Neck Surg (United States). 2015:152(1):136–42.
Luis Morejon-Troya, 1 Teodoro Maldonado Carbo Hospital, Ecuador
Joyce Jimenez-Zambrano, 1 Teodoro Maldonado Carbo Hospital, Ecuador
Mohamed Gad, Editor
About the Author: Luis Morejon-Troya completed the Otolaryngology Training Program at Universidad Estatal, Guayaquil, Ecuador in 2018. Joyce jimenez-Zambrano graduated early in 2018 from medical school at Universidad Católica de Santiago de Guayaquil, Ecuador. She is also a recipient of the Americas Committee for Treatment & Research in Multiple Sclerosis (ACTRIMS) Educational Travel Grant Award.
Correspondence: Luis Morejon-Troya, Address: Av. 25 de Julio, Guayaquil 090203, Ecuador. Email: luismorejontroy@gmail.com
Cite as: Morejon-Troya L, Jimenez-Zambrano J. Case Report: 56-year-old Woman Presenting with Progressive Hearing Loss, Chronic Otitis Media and Pulsatile Tinnitus. Int J Med Students.2019;7(1):11-14.
Copyright © 2019 Luis Morejon-Troya, Joyce Jimenez-Zambrano
International Journal of Medical Students, VOLUME 7, NUMBER 1, April 2019